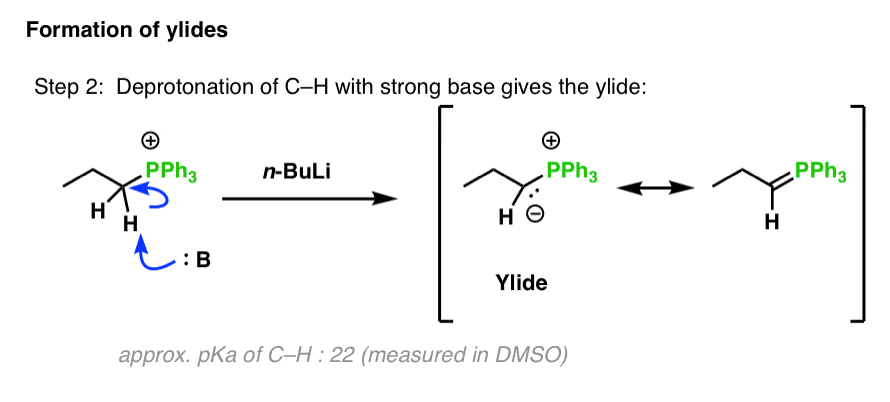
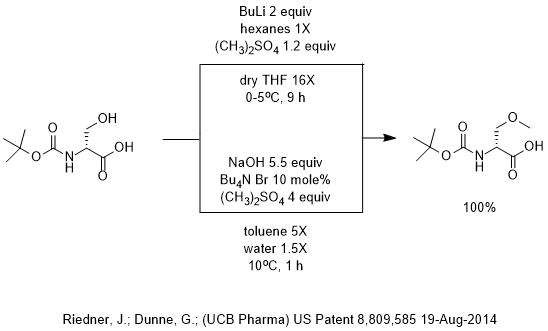
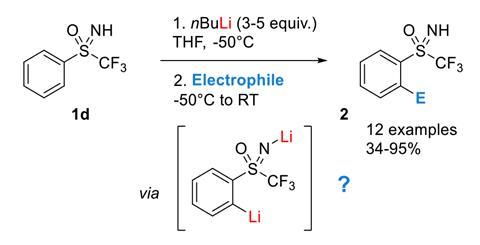
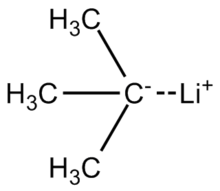
Mechanism of the Deprotonation Reaction of Alkyl Benzyl Ethers with n‐Butyllithium - Raposo - 2013 - Chemistry – A European Journal - Wiley Online Library

n-Butyllithium/N,N,N',N'-Tetramethylethylenediamine-Mediated Ortholithiations of Aryl Oxazolines: Substrate-Dependent Mechanisms | Journal of the American Chemical Society

Scheme 1. Different reactions of N, N-dimethylbenzylamine with n-BuLi /... | Download Scientific Diagram
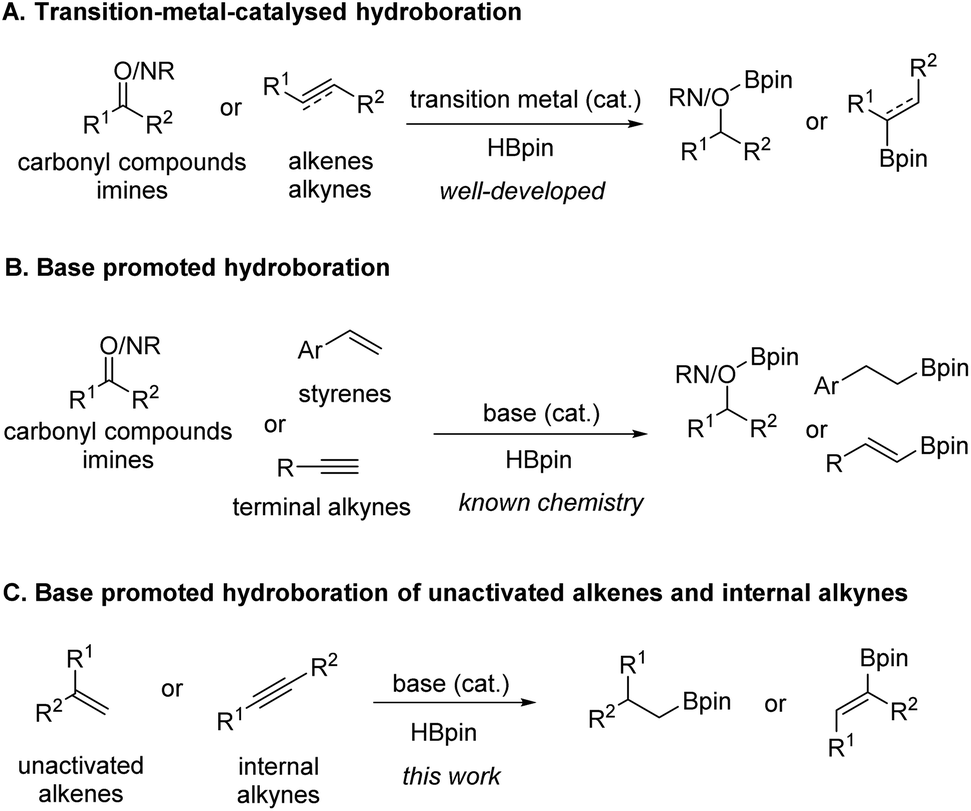
n BuLi-promoted anti -Markovnikov selective hydroboration of unactivated alkenes and internal alkynes - Organic Chemistry Frontiers (RSC Publishing) DOI:10.1039/C9QO00750D