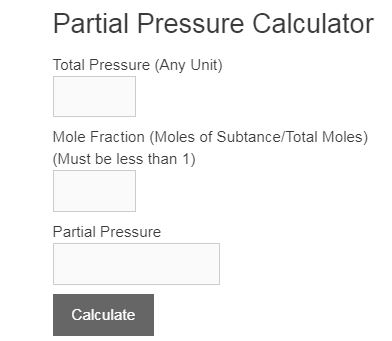
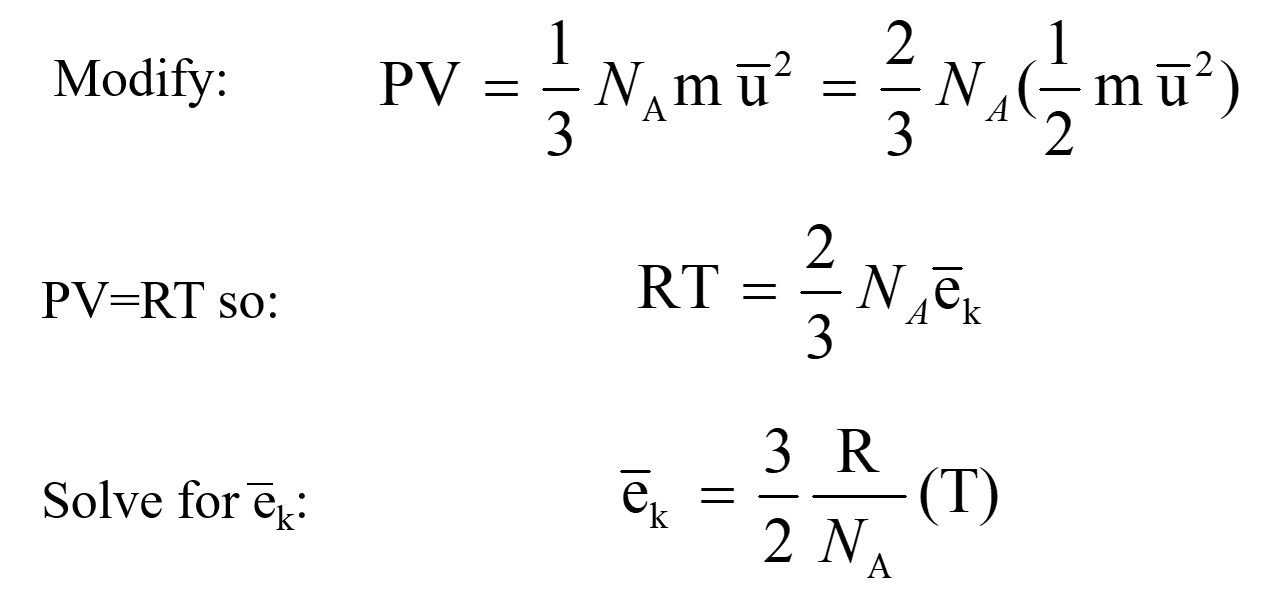A mixture of 40 g of oxygen and 40 g of helium has a total pressure of 0.9 atm. The partial pressure of oxygen is (1) 0.5 atm (2) 0.1 atm (3) 09 atm (4) 0.2 atm

calculate the total pressure in 10 litre cylinder which contain 04 gram of helium 16 gram of oxygen and 14 gram of nitrogen at 27 degree celsius also ows16022 -Chemistry - TopperLearning.com

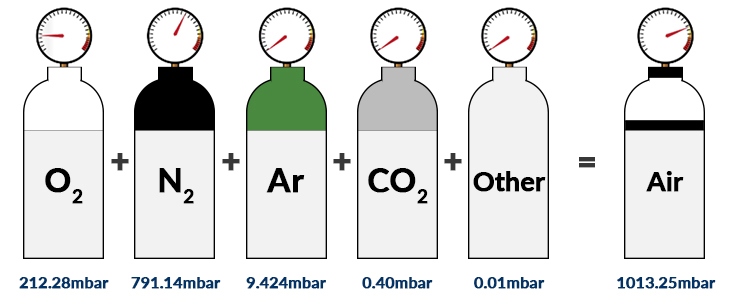
SOLVED: Calculate the partial pressure of each of the following gases in room. Air when the barometric pressure is 760 mmHg ( assume the room air contains 21% oxygen, 78% nitrogen, and

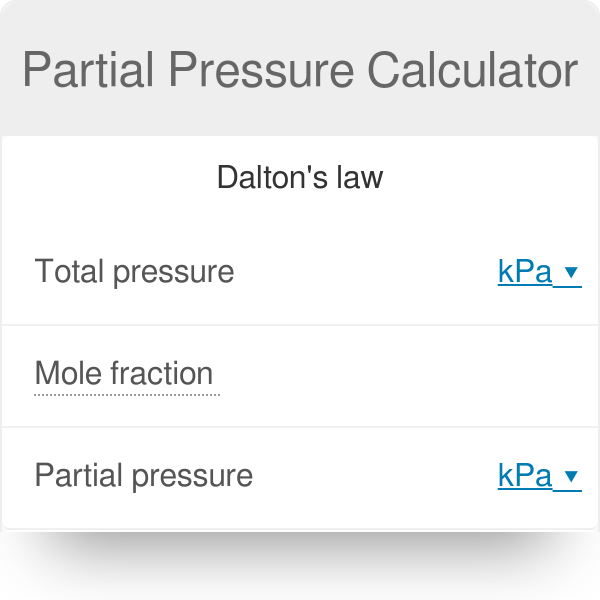
Dalton's Law of Partial Pressure: Formula | How to Find Partial Pressure - Video & Lesson Transcript | Study.com