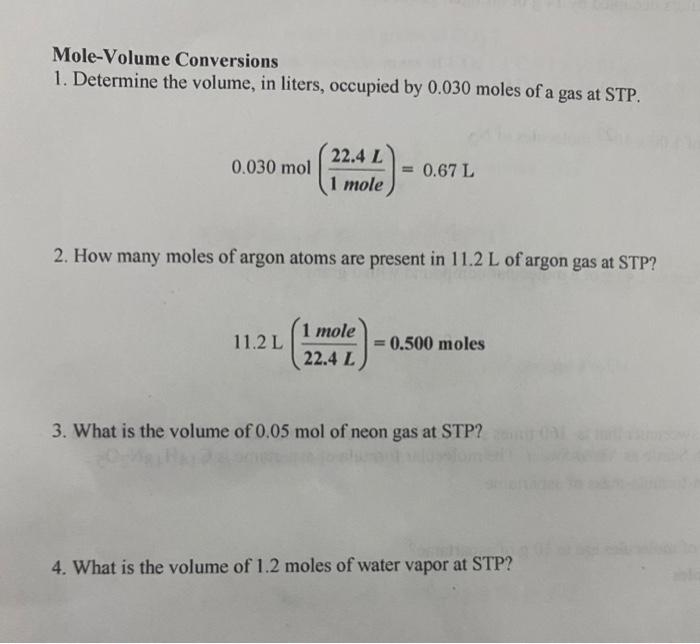SOLVED: What is the total number of moles of solute contained in 0.50 liters of 3.0M HCl? How many liters of a 0.2M NaOH solution would contain 0.4 moles of NaOH? How

Answer Key Moles & Liters volume .docx - Moles & Liters Volume We've established that it's not practical to count atoms so we often need to use | Course Hero
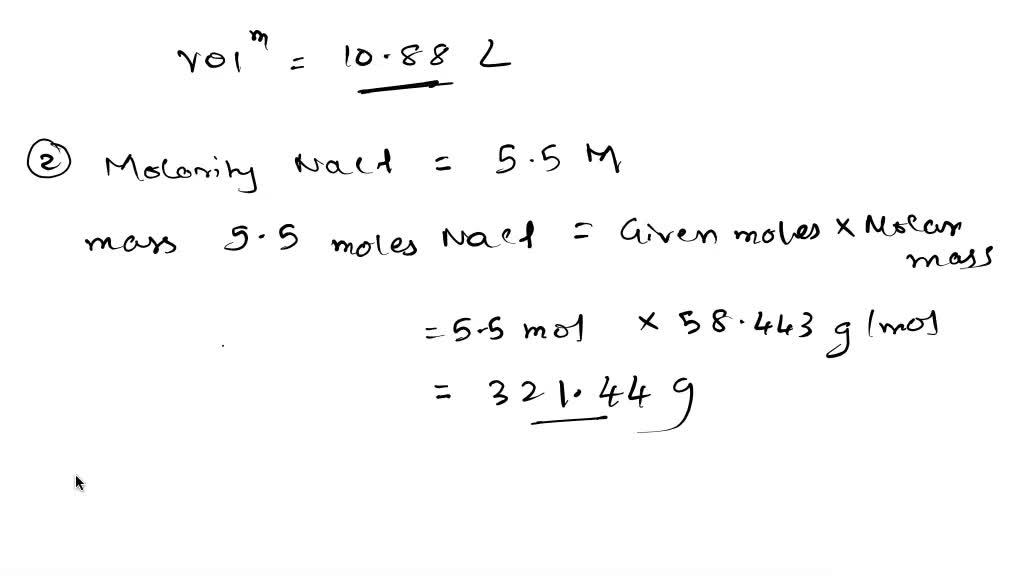
SOLVED: How many liters of a 0.114 M NaOH solution contains 1.24 moles of NaOH? The concentration of an NaCI solution (in water) expressed as Molarity is 5.5 M Express this concentration