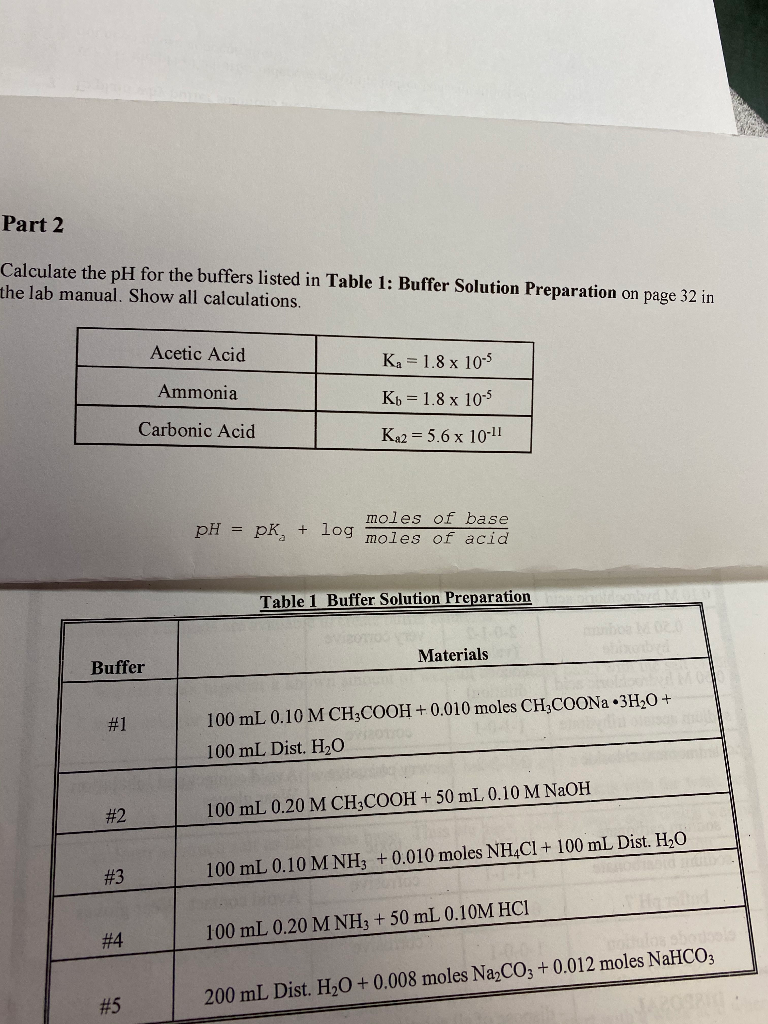
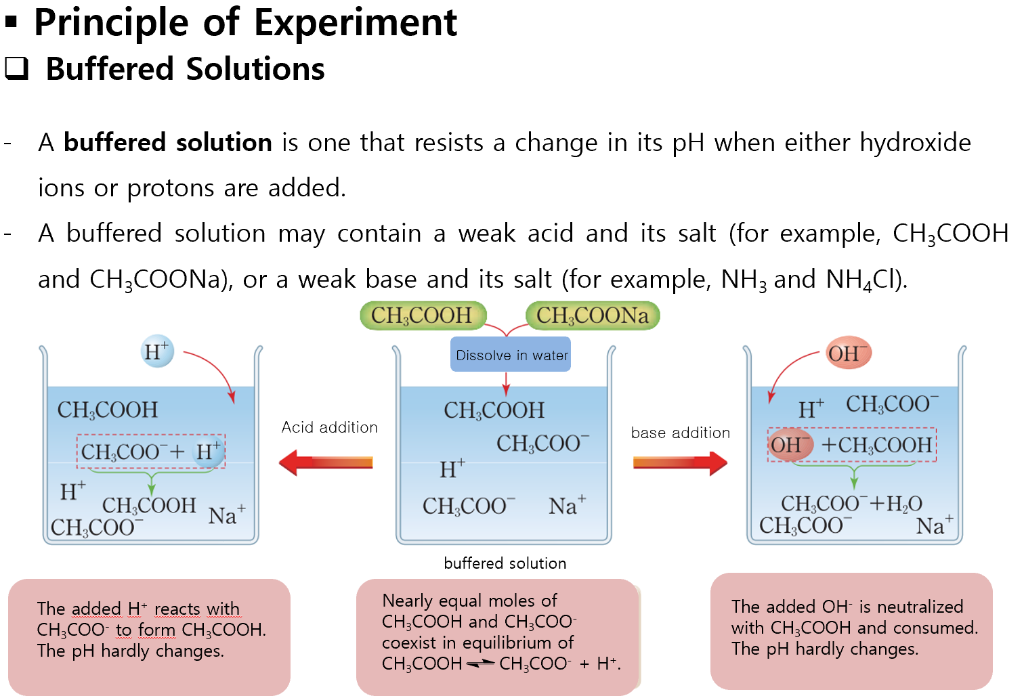
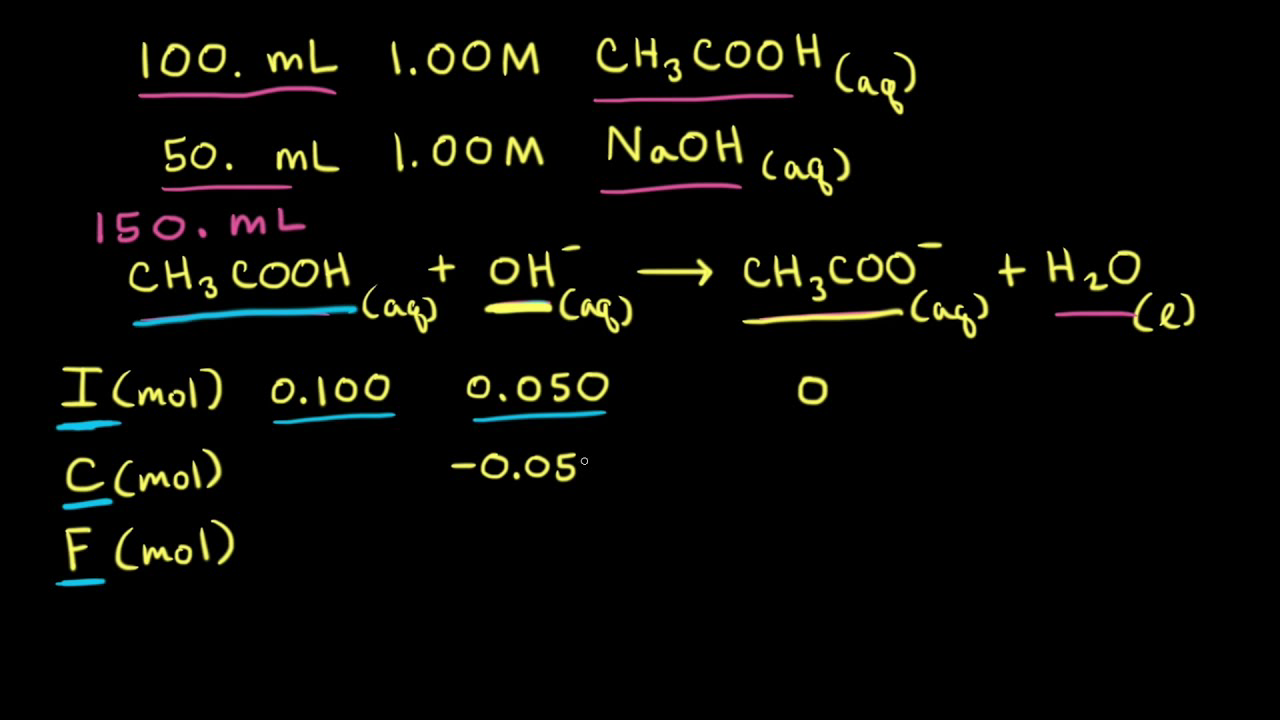
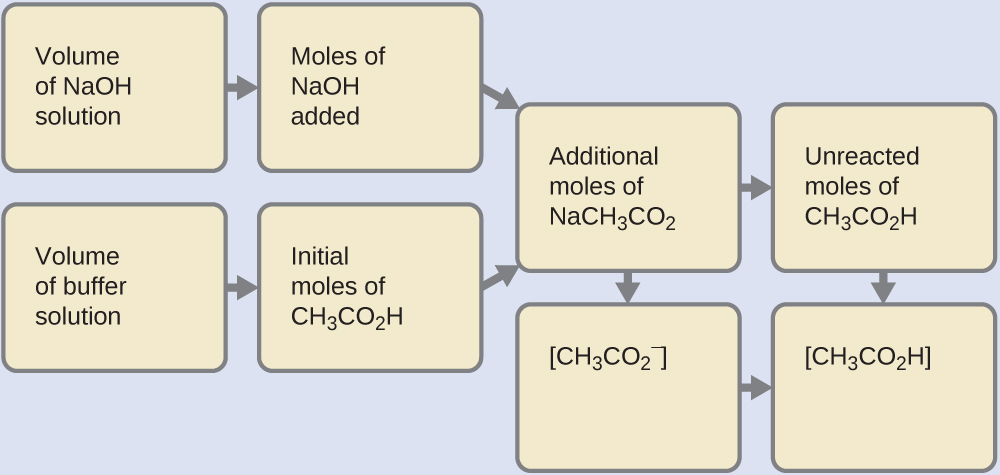
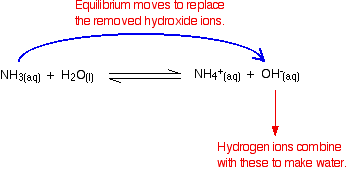
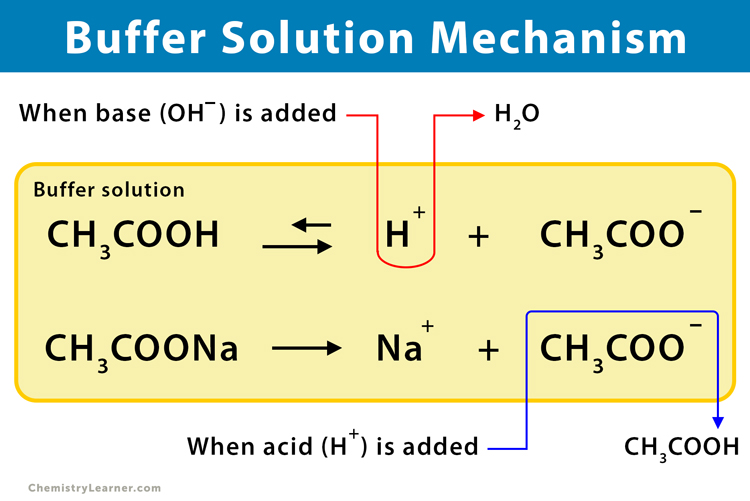
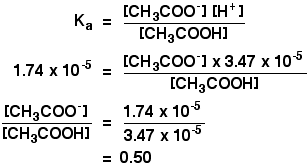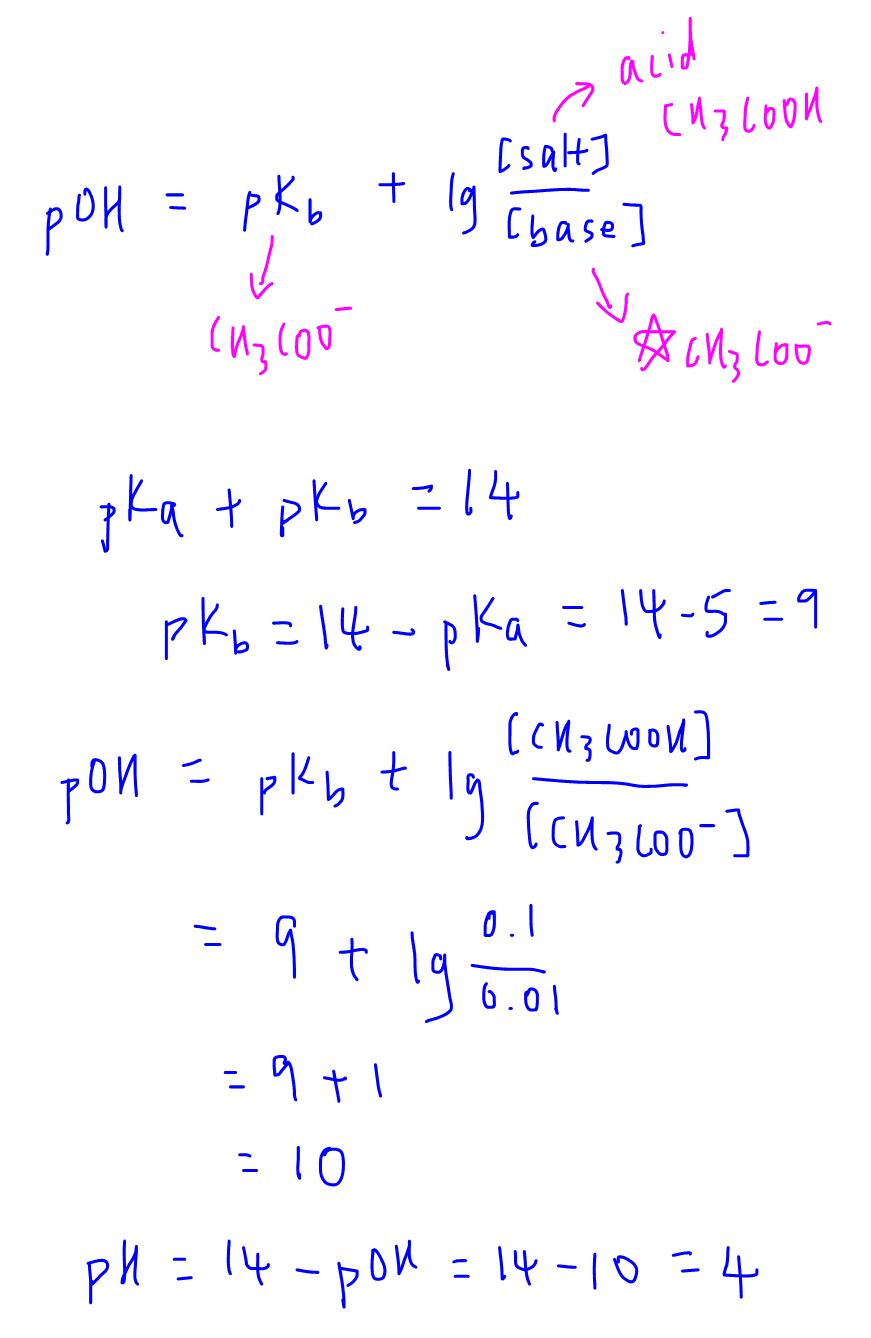A buffer solution was prepared by dissoving 0.02 mol propionic acid and 0.015 mol sodium propionate in enough water to make 1.00L of solution. (Ka for propoinic acid is 1.34 × 10^-5 ).

SOLVED: Part 4: Making an acetate buffer Reagents and glassware: SOmL 0.OM acetic acid Calculated mass of sodium acetate (which may be sodium acetate trihydrate) this mass will be less than IOg